Iron, symbol Fe, has a Body Centered Cubic structure and Gray color. 
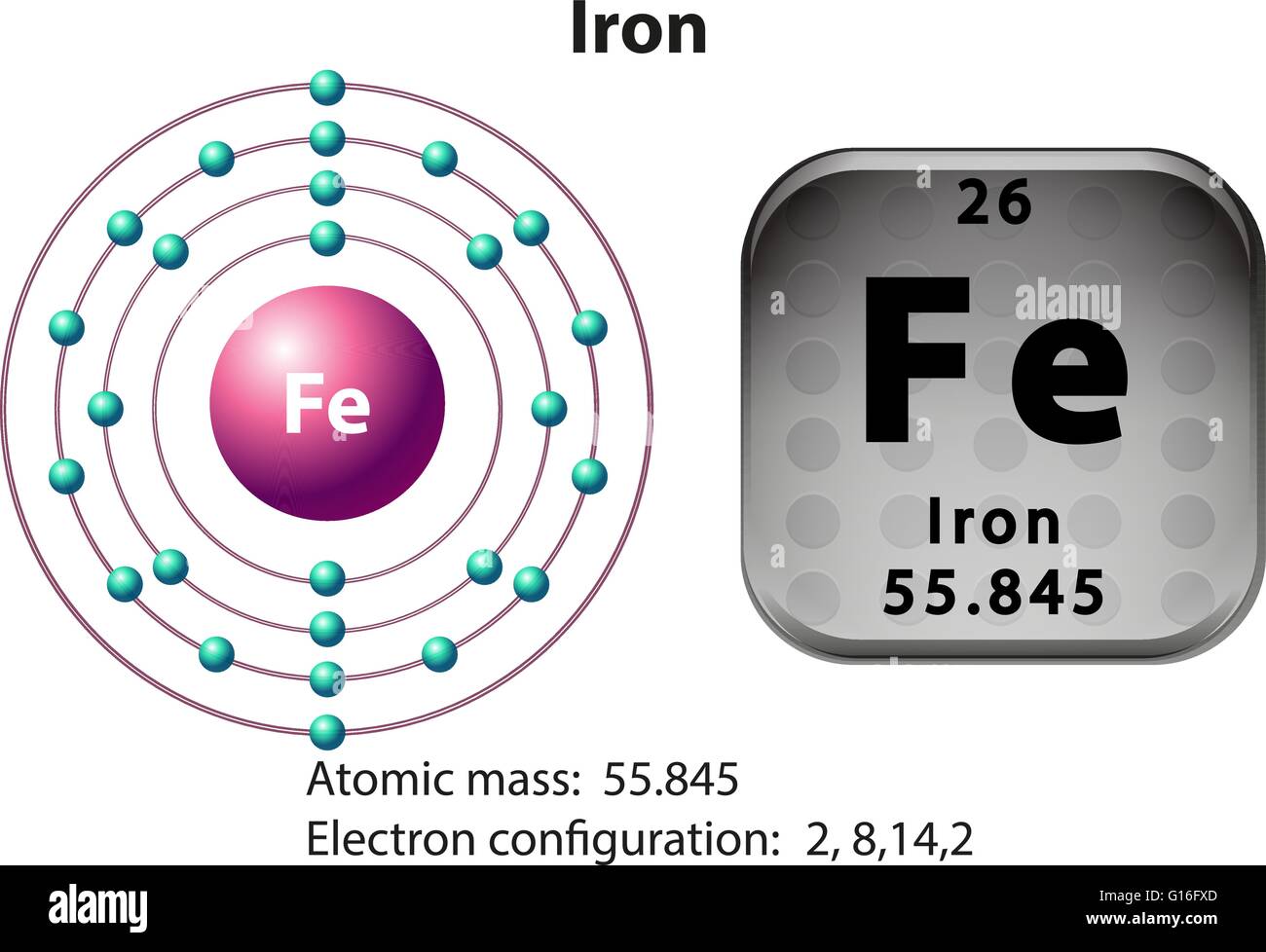
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. Isotopes of iron Naturally occurring iron ( 26 Fe) consists of four stable isotopes: 5.845 of 54 Fe (possibly radioactive with a half-life over 4.4 × 1020 years), 4 91.754 of 56 Fe, 2.119 of 57 Fe and 0.286 of 58 Fe. General Chemistry: An Atoms First Approach Unit 1: Atomic Theory Unit 2: Molecular Structure Unit 3: Stoichiometry Unit 4: Thermochem & Gases Unit 5: States of Matter Unit 6: Kinetics & Equilibria Unit 7: Electro & Thermo Chemistry Unit 8: Materials Learning Objectives To calculate the molecular mass of a covalent compound. Element 26 of Periodic table is Iron with atomic number 26, atomic weight 55.845. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. For instance, the atomic mass of 54 Fe is 53.9396147 u (9 significant digits). The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. Iron, with four naturally occurring isotopes (54 Fe, 56 Fe, 57 Fe, and 58 Fe), is no exception. The atomic radius of Iron atom is 132pm (covalent radius). As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. As mentioned in the tutorial on Atomic Mass, the atomic masses of various isotopes can be determined to a very high degree of accuracy using mass spectrometry. These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. atomic number26 atomic weight55.847 melting point1,538 C (2,800 F) boiling point3,000 C (5,432 F) specific gravity7.86 (20 C) oxidation states+2, +3, +4, +6 electron configurationAr3d64s2 Iron makes up 5 percent of Earth’s crust and is second in abundance to aluminum among the metals and fourth in abundance behind oxygen, silicon. You will need to refer to a periodic table for proton values.For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. 
A neutron is a slightly heavier particle with a mass 1.0087 amu and a charge of zero as its name suggests, it is neutral. A proton has a mass of 1.0073 amu and a charge of 1+. Neutron number plus atomic number equals atomic mass number: N+ZA. The fundamental unit of charge (also called the elementary charge) equals the magnitude of the charge of an electron (e) with e 1.602 × 10 19 C. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. In this notation, the atomic number is not included. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Iron are 56 57 58. Symbol-mass format for the above atom would be written as Cr-52. This compound is also known as Ferric Oxide or Hematite or Iron (III) Oxide. The elements whose atomic masses are written in bracket ( ) are the synthetic elements and their atomic masses values represent the Atomic Mass of the most stable isotope. atomic number26 atomic weight55.847 melting point1,538 C (2,800 F) boiling point3,000 C (5,432 F) specific gravity7.86 (20 C) oxidation states+2, +3, +4, +6 electron configurationAr3d64s2 Iron makes up 5 percent of Earth’s crust and is second in abundance to aluminum. Note: The Atomic masses are represented in the Atomic mass unit (u). The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. iron (Fe), chemical element, metal of Group 8 (VIIIb) of the periodic table, the most-used and cheapest metal. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Atomic mass of all elements (along with the rounded off values) is mentioned in the chart below. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. Physical properties The Latin name for iron is ferrum, which is the source of its atomic symbol, Fe atomic number 26 atomic weight 55.845 melting point about 1,535C boiling point about 2,750C and specific gravity of 7.87 at 20C. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
